Phonegap Healthcare Adapter Bluetooth Spec
Revision as of 15:26, 17 August 2012 by Cwdesautels (talk | contribs)
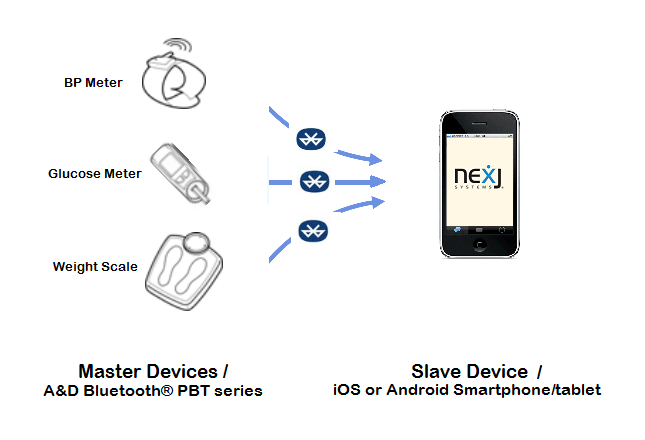
NexJ Medical Peripheral Mobile Adapter Will be designed to enable NexJ's Mobile Healthcare solutions to interact with Bluetooth peripherals.
- Main article: Phonegap Healthcare Adapter
Contents
Bluetooth Details
Bluetooth Profile
Serial Port Profile (SPP).
Bluetooth protocols
RFCOMM and Service Discovery protocols.
Pairing
Secure Simple Pairing.
Data transmission
Measurement date/time, measurement values, Bluetooth Id of remote unit, mode, and serial number of A&D PBT Series.
Master devices
A&D Bluetooth® devices PBT series, including blood pressure meter, blood glucose meter, and weight scale.
Slave devices/Access Points
mobile devices, including iOS and Android smartphones/tablets.
Process Description of Bluetooth Communication
Process Description for unpaired devices
- Enable Bluetooth on the mobile device and set the device to discoverable mode (as slave device).
- Make a measurement on selected A&D Bluetooth device such as weight scale. Upon the completion of measurement, the device (as master device) searches slave devices and initials Bluetooth communication.
- The mobile device receives the signal of the A&D Bluetooth device and prompts user to enter a PIN/passkey.
- Once the PIN is matched, the A&D Bluetooth device sends the measurement data to the mobile device using the specified format such as weight scale packet followed by the Confirmation Packet Response.
- Upon the success of connection, the two devices are paired. Then PIN code is no longer needed afterward.
Process Description for paired devices
- Enable Bluetooth on the mobile device and set the device to discoverable mode.
- Make a measurement on selected A&D Bluetooth device. Upon the completion of measurement, the A&D Bluetooth device checks its memory for previously paired address of mobile device and directly sends the measurement data to the mobile device using the specified format, followed by the Confirmation Packet Response.
Communication Packet
Communication packet consists of two sections: the dataheader section and the data section.
Header Section
| Offset | Type | Field |
|---|---|---|
| 0 | Integer (2 byte) | Packet type (always equal to 2) |
| 2 | Integer (4 byte) | Packet length (exclusive of 60 byte of data header) |
| 6 | Integer (2 byte) | Device type – 767 or 02FF (Hex) for UA-767PBT, 321 or 0141 (Hex) for UC-321PBT |
| 8 | Byte | Flag |
| 9 | Integer (2 byte) | Year of measurement |
| 11 | Byte | Month of measurement |
| 12 | Byte | Day of measurement |
| 13 | Byte | Hour of measurement |
| 14 | Byte | Minute of measurement |
| 15 | Byte | Second of measurement |
| 16 | Integer (2 byte) | Year of transmission |
| 18 | Byte | Month of transmission |
| 19 | Byte | Day of transmission |
| 20 | Byte | Hour of transmission |
| 21 | Byte | Minute of transmission |
| 22 | Byte | Second of transmission |
| 23 | Byte (6) | Bluetooth ID of remote unit |
| 29 | Byte (6) | Bluetooth ID of access point (null) |
| 35 | Byte (12) | Serial Number of A&D PBT Series |
| 47 | Byte (10) | Reserved |
| 57 | Byte | Device Battery Status (Note 1) |
| 58 | Byte | Reserved |
| 59 | Byte | Device Firmware Revision and Hardware Revision (Note 2) |
Data Section
Blood Pressure Device Specification
| Offset | Type | Field |
|---|---|---|
| 0-1 | Printable Hex | Valid (= “80”) Non-“80” means invalid BP measurement. |
| 2-3 | Printable Hex | Systolic – Diastolic |
| 4-5 | Printable Hex | Diastolic |
| 6-7 | Printable Hex | Pulse rate per minute |
| 8-9 | Printable Hex | Mean Arterial Pressure (MAP) |
Glucose Device Specification
unknown
Weight Scale Specification
Kilogram Mode
| S | T | , | + | 1 | 2 | 5 | . | 1 | 0 | k | g | <CR> | <LF> |
Pound Mode
| S | T | , | + | 0 | 4 | 0 | 5 | . | 2 | l | b | <CR> | <LF> |